Sudden unexplained death in individuals with mental health problems was first described in 1849 and a link with antipsychotic drugs was postulated 40 years ago (Reference Reilly, Ayis and FerrierReinert & Hermann, 1960; Reference Titier, Canal and DeridetTitier et al, 2005). Recently regulatory authorities in several countries have expressed concerns about a relationship between antipsychotics and the risk of QTc interval prolongation, serious arrhythmias and sudden cardiac death (Reference Citrome, Brook and WarringtonCommittee on Safety of Medicines, 1990; 1996; Reference Reinert and HermannRoyal College of Psychiatrists, 1997; National Institute for Clinical Excellence, 2004).
The granting of a licence in Europe or the USA has been prevented or delayed for several anti-psychotics and others have been withdrawn. In 1990, the Committee on Safety of Medicines (CSM) in the UK reported 13 cases of sudden unexpected death associated with the use of pimozide. This led to its restriction to a maximum daily dose of 20 mg and the recommendation of periodic electrocardiogram (ECG) monitoring of those receiving more than 16 mg daily (Reference Citrome, Brook and WarringtonCommittee on Safety of Medicines, 1990). In 1998, the CSM found evidence linking sertindole with 13 cases of serious but non-fatal arrhythmia (36 fatal cases were suspected), prompting Lundbeck to voluntarily suspend sale of the drug (Committee on Safety of Medicines, 1998). Interestingly, in 2001 it was reintroduced by the European Committee of Proprietary Medicinal Products on a named-patient basis. In 1998, following concerns over QTc prolongation, thioridazine, mesoridazine and droperidol received a black-box warning in the USA and ziprasidone received a lower bold-print warning. By the end of 2000, the deaths of 21 people prescribed thioridazine had come to the attention of the CSM (Reference MaronMedicines and Healthcare Products Regulatory Agency, 2000), leading to its restriction to second-line treatment of schizophrenia in people under the direct care of a consultant psychiatrist. Shortly afterwards droperidol was withdrawn from the market by the manufacturers. The launch of the atypical anti-psychotic ziprasidone continues to be delayed in the UK because more than 10% of patients taking the drug developed modest QTc interval prolongation (Box 1). However, in one large series of patients taking ziprasidone, only 2 in 2988 individuals had QT intervals longer than 500 ms (Trenton et al, 2003).
Box 1 QTc intervals
The QTc interval measures the time between the start and end of electrical ventricular activity. It is obtained by correcting the QT for heart rate using a regression formula (e.g. Bazett, Friderica). See part 2 of this overview (Reference Abdelmawla and MitchellAbdelmawla & Mitchell, 2006) for further discussion.
Several authors have asked whether these restrictions are justified, arguing that the relative and absolute risks of serious adverse events are not clear from case reports or ECG changes alone (Reference de Bruyne, Hoes and KorsDershwitz, 2002).
In this article we examine the possible mechanisms of antipsychotic-induced sudden death and the likelihood of serious adverse events in those with drug-induced ECG changes.
What is meant by sudden death?
Sudden death refers to the unexpected death of a person who has no known acutely life-threatening condition and yet dies of a fatal medical cause (Box 2). The incidence of sudden death in the population is about 400 000 cases per year in the USA and perhaps 80 000 per year in the UK (Reference Malik and CammMaron, 2003). Yet it is rare in psychiatric settings, primarily because of the relatively short periods of observation compared with the lifetime risk. Mehtonen et al(1991) found only 49 sudden deaths among 25 000 people with psychiatric disorders followed for 3 years. Reference Jones, Macias and BarreiraJusic & Lader (1994) emphasised that most sudden deaths in psychiatric settings are unexplained by post-mortem examination.
Box 2 Causes of sudden cardiac death
-
• Coronary artery disease
-
• Ischaemic cardiomyopathy
-
• Non-ischaemic cardiomyopathy
-
• Hypertrophic cardiomyopathy
-
• Arrhythmogenic right ventricular cardiomyopathy
-
• Commotio cordis
-
• Sarcoidosis
-
• Amyloidosis
-
• Myocarditis
-
• Valvular heart disease
-
• Congenital heart disease
-
• Cardiac tumours
-
• Long QT syndrome
-
• Brugada syndrome (Box 3)
-
• Wolff–Parkinson–White syndrome (Box 3)
-
• Electrolyte abnormalities
-
• Thyrotoxicosis
-
• Pro-arrhythmia from anti-arrhythmic agents
-
• Cocaine
A modest-sized study of 74 cases of sudden death from five psychiatric hospitals in the north-east of England over a 12-year period generated some useful epidemiological information (Reference Reilly, Ayis and FerrierReilly et al, 2002). Sudden death accounts for about 5% of all deaths in psychiatric hospitals. Around 20% of sudden deaths of people with mental health problems occur in those under 65. Contrary to early fears, only very rarely do deaths occur during restraint, behavioural disturbance or shortly after rapid tranquillisation. Antipsychotics are the most commonly prescribed drug in reported cases of sudden death, but in this series of 74 deaths, 40% of the individuals were not prescribed antipsychotics at the time of the event. About a third of individuals have schizophrenia, a third have a mood disorder and a third have an organic diagnosis (such as delirium or dementia).
Witnessed accounts suggest that sudden death is heralded by abrupt loss of consciousness within 1 h of the onset of acute symptoms. The symptoms can be of chest pain, weakness or fatigue, and palpitation. If there are no witnesses, it has been suggested that the description of sudden death may be appropriate if an individual known to have been medically stable less than 24 h previously dies without clinical evidence of a cause of death (Reference Moss, Schwartz and CramptonMyerberg, 1997; Reference Ösby, Correia and BrandtPriori et al, 2001). In the sudden death of young people, post-mortem examination reveals a pre-existing cardiac cause in most cases (often called sudden cardiac death). Yet in the majority of cases of sudden death of young people with mental health problems there is thought to be no structural abnormality, although no good postmortem series in psychiatric cases has been reported (Reference Reilly, Ayis and FerrierReilly et al, 2002; Reference Malik and CammMaron, 2003).
Box 3 Brugada and Wolff–Parkinson–White syndromes and commotio cordis
Brugada syndrome
This is an inherited form of cardiac arrhythmia, presenting with an ECG pattern of S–T segment elevation in leads V1–V3, and incomplete or complete right bundle branch block. In about 20% of cases, the underlying cause is a genetic defect in the SCN5A gene
Wolff–Parkinson–White syndrome
The syndrome is characterised by an abnormal band of atrial tissue which connects the atria with the ventricles and can electrically bypass the atrioventricular node. As this node and bypass tract have different conduction speeds and refractory periods, a re-entry circuit can develop, causing paroxysms of tachycardia
Commotio cordis syndrome
In commotio cordis a blunt non-penetrating impact to the chest causes cardiac arrest
In one of the few autopsy series of sudden cardiac death in 14 patients taking antipsychotics, Reference Glassman and BiggerGreen (1988) reported the cause of death as ‘chronic cardiac toxicity’. In a survey of autopsies associated with psychotropic medication in Finland, of the individuals who succumbed to sudden cardiac death despite structurally normal hearts, half had been entirely asymptomatic previously (Chugh et al, 2000). This suggests that arrhythmias are responsible in most cases rather than one of a myriad of other possible causes such as hypertrophic cardiomyopathy, commotio cordis (Box 3), coronary artery anomalies, ruptured aortic aneurysm, myocarditis and valvular heart disease (Reference Saz and DeweySpector, 2005). The true distribution of initial arrhythmias is not known, since several minutes usually elapse between collapse and rhythm assessment. Reference DuBuskeEngdahl et al(2002) reported that recordings obtained during spontaneous episodes of sudden cardiac death demonstrated ventricular arrhythmias in about 85% of cases.
How common is antipsychotic-induced sudden death?
Mortality in people with schizophrenia
There is an excess mortality rate (standardised mortality ratios for all causes of death are 1.7 times higher in men and 1.3 times higher in women) in people with psychiatric illnesses, even after allowing for the effect of suicide (Reference Trenton, Currier and ZwemerWaddington et al, 1998; Reference Ogata and Narahashi…sby et al, 2000). This non-suicide elevated risk holds true for people with schizophrenia (Reference Allebeck and WistedtAllebeck & Wistedt, 1986; Reference Brown, Barraclough and InskipBrown et al, 2000) or depression (Reference Sadanaga, Sadanaga and YaoSaz & Dewey, 2001), as well as for those with delirium and dementia. The standardised mortality ratio is greater than 2 for both males and females with schizophrenia (Reference Hennessy, Bilker and KnaussHewer & Rossler, 1997; Reference Ogata and Narahashi…sby et al, 2000).
The causes are complex and include an excess of physical health problems, an increased rate of vascular risk factors (smoking, lack of exercise, obesity, diabetes) and a lower than expected uptake of healthcare services (Nose et al, 2003; Connolly & Kelly, 2005). However, the largest single cause is cardiovascular disease. When examined in detail, data show that people with schizophrenia have an increased incidence of arrhythmia, syncope and heart failure, diabetes and stroke (Reference Connolly and KellyCurkendall et al, 2004).
Two major risk factors for excess mortality in psychiatric patients are medical comorbidity and treatment with antipsychotic medication. The latter is not necessarily causative, as higher doses might be linked with mortality through a confounding factor such as worse physical health in those with more serious mental health problems. Compared with the overall excess of non-suicide mortality, the number of deaths that might be attributable to medication is small, but most argue that iatrogenic mortality must be taken seriously, with appropriate steps taken towards prevention (Reference Abdelmawla and MitchellAbdelmawla & Mitchell, 2006).
Mortality in patients prescribed antipsychotics
Reference Rautaharju and ZhangRay et al(2001) conducted a retrospective cohort study of half a million Medicaid patients in the US state of Tennessee between 1988 and 1993, before the introduction of atypical antipsychotics. They found that the risk of sudden death for individuals receiving antipsychotic drugs was 2.39 times that for ‘non-users’. Reference Montanez, Ruskin and HebertMontout et al(2002) conducted a 4-year prospective study of 3474 individuals with schizophrenia. The risk of ‘all-cause’ death and the risk of ‘non-suicide cause’ death was increased in users of thioxanthenes (alone or with other drugs) and atypical antipsychotics (odds ratio OR = 2.06). Reference Harrigan, Miceli and AnzianoHennessy et al(2002) retrospectively examined the rates of cardiac arrest and ventricular arrhythmia in people with treated schizophrenia and in non-schizophrenia controls in a cohort of 90 000 obtained from three US Medicaid programmes between 1993 and 1996. The antipsychotics used included clozapine, haloperidol, risperidone and thioridazine. People treated for schizophrenia had higher rates of cardiac arrest and ventricular arrhythmia than controls. The relative risk for cardiac arrest and ventricular arrhythmia in those taking risperidone compared with haloperidol was 1.5, but there was no dose–response relationship. Reference Lin, Chen and WangLiperoti et al(2005) conducted a case–control study of residents of nursing homes in six US states. Use of conventional antipsychotics (but not atypical antipsychotics) was associated with a nearly twofold increase in risk of hospitalisation for ventricular arrhythmias or cardiac arrest, particularly in those with pre-existing cardiac disease.
Mortality following rapid tranquillisation
Concern has been expressed regarding reports of sudden death after the emergency use of psychotropic medication. Although this is sometimes linked with rapid tranquillisation in the literature on acute behavioural disturbance, there have been no deaths or serious cardiovascular events recorded during several recent large trials involving the treatment of over 1500 patients with ziprasidone v. haloperidol, olanzapine v. haloperidol or midazolam v. haloperidol–promethazine mix (TREC Reference Titier, Girodet and VerdouxCollaborative Group, 2003; Reference Chugh, Kelly and TitusCitrome et al, 2004)
In the Finnish study of 49 cases of sudden cardiac death (Mehtonen et al, 1991) none was associated with a struggle and most patients died in bed more than 4 days after admission, suggesting little relationship with acute behavioural disturbance. Indeed in the literature there are few cases of sudden death that are clearly linked with rapid tranquillisation itself as opposed to cumulative antipsychotic dosing.
Reference Jones, Macias and BarreiraJusic & Lader (1994) reported two such cases. In the first, a 31-year-old man died within minutes of being forcibly given intramuscular medication. High concentrations of fluphenazine decanoate were found in the heart, suggesting that the intramuscular depot was inadvertently given intravenously. In the second case, a 27-year-old man with schizophrenia was given intravenous haloperidol and diazepam at appropriate doses and suffered a cardiac arrest but showed no pathology at post-mortem examination. However, caution must be taken when interpreting post-mortem drug concentrations.
Altimari and colleagues, for the National Alliance for the Mentally Ill, compiled a list of 142 deaths over a 10-year period that had occurred while patients were in restraint or seclusion. From these and other data (Reference Myerberg and BraunwaldNational Alliance for the Mentally Ill, 1998), it is apparent that in some cases the mechanism of sudden death following rapid tranquillisation appears to involve inadvertent suffocation rather than a drug-related effect.
ECG irregularties and mechanisms of sudden death
Considerable research has attempted to clarify the underlying electrical substrate that predisposes individuals to ventricular fibrillation and sudden cardiac death. It is known that ventricular tachycardia degenerates first to ventricular fibrillation or torsade de pointes and then quickly to asystole. Most work has concentrated on risk in those who have ischaemic heart disease or other causes of heart failure (Reference Kriwisky, Perry and TarchitskyLane et al, 2005). In people with structurally normal hearts only a limited number of risk factors have been studied, and most evidence relates to electrocardiographic markers such as the QT interval (for further information see part 2 of this overview: Reference Abdelmawla and MitchellAbdelmawla & Mitchell, 2006).
Torsade de pointes
Although torsade de pointes is often a transient, self-correcting condition, in about a third of cases it leads to ventricular fibrillation and about 1 in 10 events cause sudden death (Reference Obias-Manno and WijetungaO’Brien & Oyebode, 2003; Reference Modai, Hirschmann and RavaMontanez et al, 2004). Torsade de pointes is largely unpredictable but certain risk factors can be identified (Box 4). Drug-induced torsade de pointes typically arises with the use of medications that cause QT prolongation beyond 500 ms at the same time as inducing polymorphic ventricular tachycardia (twisting changes in QRS morphology on the ECG) (Reference Montout, Casadebaig and LagnaouiMorganroth, 1993; Reference Zarate, Baldessarini and SiegelZareba & Moss, 2003). Patients with QT prolongation are at particular risk if they have QT-related arrhythmias such as sudden bradycardia or long pauses (e.g. compensatory pauses after ventricular ectopy), enhanced U-waves, T-wave alternans, polymorphic ventricular premature beats, couplets or non-sustained polymorphic ventricular tachycardia. The management of torsade de pointes includes withdrawal of offending agents, empirical administration of magnesium regardless of serum magnesium, correction of serum potassium to 4.5–5 mEq/l and manoeuvres to increase heart rate (isoprenaline or pacing) if necessary. However, prevention is probably the most important strategy for at-risk patients.
Box 4 Risk factors for QT prolongation and torsade de pointes
-
• Older age
-
• Bradycardia
-
• Congenital long-QT syndrome
-
• Electrolyte abnormalities (especially hypo-kalaemia or hypomagnesaemia)
-
• Female gender
-
• Heart disease (especially left ventricular hypertrophy, ischaemia, or low left ventricular ejection fraction)
-
• Significant T-wave abnormalities
-
• Mutations of the sodium channel
-
• LQT3 mutations of the SCN5A sodium channel gene
-
• Mutations of the potassium channel gene
-
• LQT1 mutations of the KCNQ1 gene
-
• LQT5 mutations of the minK gene
-
• LQT2 mutations of the HERG gene
-
• LQT6 mutations of the MiRP1 gene
Abnormalities of the QT interval
Abnormalities in the QT interval can be divided into three types:
-
• prolongation of the interval
-
• differences in the interval across different ECG leads (QT dispersion)
-
• QT variability with time.
Only prolongation has been studied in any depth in relation to antipsychotics. Rate-corrected QT intervals (QTc) are usually around 400 ms in duration, and values lower than 440 ms are considered normal (Reference Ganguly, Kotzan and MillerGlassman & Bigger, 2001); QTc values above 450 ms (men) or 460 ms (women) are considered prolonged. Thus, there is a borderline area of uncertain significance (Reference Al-Khatib, LaPointe and KramerAl-Khatib et al, 2003; Reference Suessbrich, Schonherr and HeinemannTaylor, 2003). The cut-off point of 500 ms is widely accepted as indicating substantially higher risk of arrhythmias such as torsade de pointes (Reference BotsteinBotstein, 1993; Reference Warner, Barnes and HenryWelch & Chue, 2000; Reference Ganguly, Kotzan and MillerGlassman & Bigger, 2001; Reference Liperoti, Gambassi and LapaneMalik & Camm, 2001). Progressive lengthening of the QT interval in repeated ECGs is a warning sign (Rautaharju & Zhang, 2002). Broadly, the greater the QTc interval the higher the risk of torsade de pointes and other serious arrhythmias. Length of the QTc interval has been associated with risk of sudden death following myocardial infarction and congenital long-QT syndrome (Reference MorganrothMoss, 1993; Reference Czekalla, Beasley and Dellvade Bruyne et al, 1999). Evidence suggests that 5–10% of people in whom drug-induced torsade de pointes develops harbour genetic mutations associated with long-QT syndrome (Reference DershwitzDilaveris & Stefanadis, 2005).
Mechanism of drug-induced QT prolongation
Psychotropic drugs can affect the QT interval through blockade of ion channels, slowing depolarisation. Slow depolarisation leads to the prolongation of the QRS interval, extending the QT interval. Drugs that block sodium channels in depolarisation produce modest prolongation of the QRS complex and are not thought to be associated with sudden death in individuals without pre-existing cardiac disease. Repolarisation, on the other hand, involves sodium, calcium and potassium. The potassium channel most often involved in drug-induced QT abnormalities is the delayed rectifier channel for potassium (inward rectifier K+ channel IKr, encoded by the human ether-a-go-go-related gene (HERG) (Reference Drolet, Vincent and RailDuBuske, 1999). Drugs that block IKr channels (such as thioridazine) can slow repolarisation, which leads to the prolongation of the S–T segment and the T-wave, and therefore the QTc interval. Consequently these drugs can induce torsade de pointes and sudden death in apparently healthy adults (Reference Welch and ChueWitchel & Hancox, 2000).
In animal studies it has been shown that clozapine, haloperidol, pimozide, sertindole and thioridazine block repolarising IKr channels in vitro (Reference Studenik, Lemmens-Gruber and HeistracherSuessbrich et al, 1997; Reference Drici, Wang and LiuDrolet et al, 1999). In addition, thioridazine and pimozide appear to inhibit calcium channels in rat myocytes, while haloperidol and chlorpromazine block sodium (Na+) conduction in myocytes (Reference O'Brien and OyebodeOgata & Narahashi, 1989). Furthermore, in isolated spontaneously beating guinea pig Purkinje fibres, chlorpromazine and thioridazine induced early ‘after depolarisation’ (Reference Straus, Bleumink and DielemanStudenik et al, 1998), a hypothesised trigger for torsade de pointes (Reference Donatini, LeBlaye and KruppDrici et al, 1998). In an isolated feline heart model, haloperidol, risperidone, sertindole, clozapine and olanzapine produce a dose-dependent prolongation of the QT interval (Reference Donatini, LeBlaye and KruppDrici et al, 1998). Although helpful in providing insight into potential antipsychotic drug-induced QT prolongation in humans in vivo, it should be borne in mind that high doses of antipsychotics were used in these studies.
Drug–drug interaction in the cytochrome P450 (CYP) system is a related hazard, which we discuss in part 2 of this overview (Reference Abdelmawla and MitchellAbdelmawla & Mitchell, 2006).
What is the risk of sudden death with established ECG changes?
Early work suggested a simple relationship between degree of QT prolongation and adverse events. Some have suggested that a 5% increase in baseline risk of arrhythmic events occurs for every 10 ms increase in the QTc interval (Reference MossMoss et al, 1991). Although attractive, this is now known to be an oversimplification of a highly complex relationship between the QT interval and cardiovascular risk (Reference Cavero, Mestre and GuillonCavero et al, 2000).
Reference Modai, Hirschmann and RavaMontanez et al(2004) reviewed seven large-scale prospective cohort studies encompassing 36 031 individuals from the general population. In people with prior cardiovascular disease, QTc prolongation increased the relative risk of death two- to fourfold, but in the overall population the link between QTc prolongation and cardiovascular mortality/sudden death was inconsistent. Risk factors for sudden cardiac death in healthy people and those with cardiac failure are listed in Box 5.
Box 5 Risk factors for sudden death
Healthy people
-
• Repolarisation abnormalities (long QT syndromes)
-
• Ventricular pre-excitation (Wolff–Parkinson–White syndrome)
Those with cardiac failure
-
• Ischaemic heart disease
-
• Dilated cardiomyopathy
-
• Left ventricular systolic dysfunction
-
• Abnormal signal-averaged ECG
-
• Non-sustained ventricular tachycardias on Holter monitor recordings
-
• Reduced heart rate variability
-
• T-wave alternans
-
• B-type natriuretic peptide
After Reference Nose, Barbui and TansellaObias-Manno & Wijetunga (2004); Reference Kriwisky, Perry and TarchitskyLane et al(2005)
Adverse events associated with specific antipsychotics
ECG changes
Several observational studies suggest that people treated with antipsychotics have a higher than expected rate of ECG changes (Fig. 1). Reference Waddington, Youssef and KinsellaWarner et al(1996) found QTc prolongation in 23% of 111 in-patients with chronic schizophrenia compared with 2% of 42 controls.
Reference Ray, Meredith and ThapaReilly et al(2000) compared 339 patients taking antipsychotics (17% at high or very high doses) without atrial fibrillation or bundle-branch block with a group of 101 healthy controls. In this study QTc prolongation was found in 8% of those taking antipsychotics and 15% of those taking anti-psychotics and antidepressants in combination. Increasing the antipsychotic dose was associated with increased risk of QTc lengthening.
Reference Kalsekar, Makela and MoellerKang et al(2000) found that 13 of 54 patients treated with clozapine showed new-onset ECG abnormalities, with a dose-dependent increase in QTc interval. Reference Liberatore and RobinsonLin et al(2004) compared ECGs from 412 in-patients taking antipsychotics for schizophrenia with those of 138 controls. Logistic regression analysis showed that significant predictors for QTc prolongation were: older age, female gender and increased doses of clozapine, chlorpromazine, thioridazine and sulpiride. In a prospective study Reference Hägg, Spigset and BateHarrigan et al(2004) randomised 160 patients with psychotic disorders to either haloperidol, thioridazine, ziprasidone, quetiapine, olanzapine or risperidone. Mean QTc intervals were prolonged but did not exceed 500 ms in any individual, and interestingly were not affected by adding a CYP inhibitor.
Sadanaga et al(2004) examined a cohort of 688 patients prescribed psychotropic drugs and observed a QTc interval >460 ms in 96 (14%). However, they also monitored 97 individuals using a 24-h ECG and estimated the QT intervals at heart rates of 80 and 50 beats/min. The QT interval at a heart rate of 80 beats/min exceeded 400 ms in 14 (15%) patients and at the heart rate of 50 beats/min it exceeded 480 ms in an additional 12 (12%) patients.
Pooled data from four randomised clinical trials involving 2700 patients did not reveal any ECG abnormalities due to olanzapine (Reference Curkendall, Mo and GlasserCzekalla et al, 2001).
Serious cardiac events and sudden death
Case reports
Case reports linking antipsychotic drugs and cardiac arrhythmia have appeared for thioridazine (Reference Lane, Cowie and ChowLiberatore & Robinson, 1984; Reference Domino, Anderson and PolissarDonatini et al, 1992), pimozide (Reference Citrome, Brook and WarringtonCommittee on Safety of Medicines, 1990), sulpiride, droperidol, haloperidol (Reference Kopecky and GershKriwisky et al, 1990; Reference Hoehns, Stanford and GeraetsJackson et al, 1997), sertindole (Committee on Safety of Medicines, 1999), risperidone (Reference Witchel and HancoxZarate et al, 1997), ziprasidone (Reference Adolfsson and LindblomAdolfsson & Lindblom, 2002) and clozapine (Reference Kao, Kirk and EversKillian et al, 1999; Reference GreenHägg et al, 2001). Clozapine has also been linked with potentially fatal myocarditis and cardiomyopathy in physically healthy young adults with schizophrenia. Active myocarditis is suspected as a common precursor of idiopathic dilated cardiomyopathy but is found in a minority of patients with dilated cardiomyopathy (Reference Killian, Kerr and LawrenceKopecky & Gersh, 1987). Confirmatory evidence can be obtained only from endomyocardial biopsy or autopsy.
Since 1980 at least 213 cases of myocarditis in people taking clozapine have been reported in the literature, with at least 50 fatalities (Reference GreenHägg et al, 2001; Reference Mehtonen, Aranki and MalkonenMerrill et al, 2005). In some series about half of cases of clozapine-induced myocarditis are fatal, and about a quarter of these people die suddenly.
Overdose studies
In overdose there may be a dose–response relationship between thioridazine and increasing QTc interval that is not seen for chlorpromazine (Reference SpectorStrachan et al, 2004). However, a case series has linked chlorpromazine with ventricular arrhythmias and torsade de pointes (Reference Hewer and RosslerHoehns et al, 2001). Reference Buckley, Whyte and DawsonBuckley et al(1995) showed that cardiotoxicity (as indicated by tachycardia, prolonged QT and QTc intervals, widened QRS complex and arrhythmias) appears to be particularly common following thioridazine overdose (Reference Buckley, Whyte and DawsonBuckley et al, 1995). A review of case reports of overdose of antipsychotics, including the atypicals olanzapine, quetiapine and amisulpride (but not aripiprazole or zotepine), revealed fatal outcomes for nearly all of the typical antipsychotics (Trenton et al, 2003). However, Capel and colleagues found no fatalities in a case series of overdoses of olanzapine, clozapine, risperidone and sulpiride over a 7-month period (Reference Capel, Colbridge and HenryCapel et al, 2000).
Epidemiological studies
Case reports are useful indicators of a potential problem, but estimation of the magnitude of risk requires large-scale experimental or pharmaco-epidemiological studies. In a prospective cohort study of 88 in-patients with schizophrenia who had been followed for 10 years, Reference Trenton, Currier and ZwemerWaddington et al(1998) showed a relationship between increasing number of antipsychotic drugs prescribed concurrently and shorter survival (RR = 2.46). Reference Ray, Meredith and ThapaReilly et al(2000) in a study of ECGs from 495 psychiatric patients found evidence of QTc prolongation with thioridazine (OR = 5.3) and droperidol (OR = 6.7). They found a positive dose–response relationship between anti-psychotic and QTc prolongation. Risk was elevated if patients were prescribed more than 1000 mg chlorpromazine equivalent per day (the Reference Reinert and HermannRoyal College of Psychiatrists (1997) advises caution with doses above this level). Reference Merrill, Dec and GoffModai et al(2000) found a fourfold increase in sudden death among 561 individuals taking clozapine v. 4918 controls and in 75% of the deaths there was no known medical cause.
Two American teams studied administrative databases (Reference Rautaharju and ZhangRay et al, 2001; Reference Harrigan, Miceli and AnzianoHennessy et al, 2002). In the Hennessy et al study, the team compared the relative risk of cardiac arrest or ventricular arrhythmia and death in patients on clozapine, risperidone, and thioridazine with the risk in those on haloperidol. Clozapine and thioridazine were not associated with a higher risk of adverse cardiac events than haloperidol, whereas risperidone was associated with a higher risk of arrhythmic events and all-cause mortality than haloperidol. In addition, there was a dose-dependent relationship between thioridazine and arrhythmic events, particularly at doses of more than 600 mg (Reference Harrigan, Miceli and AnzianoHennessy et al, 2002).
More recently, researchers from The Netherlands (Reference Strachan, Kelly and BatemanStraus et al, 2004) performed a population-based case–control study of 554 cases of sudden cardiac death. Risk of an event was three times higher in those receiving antipsychotics, six times higher in those receiving butyrophenones (predominantly pipamperone and haloperidol) but not significantly higher in those receiving phenothiazines (predominantly chlorpromazine and thioridazine). The association was also dose- and duration-related and there was a trend for risk to be greater in those under 65 years of age.
Following concerns about droperidol in the USA, Reference Dilaveris and StefanadisDomino et al(1999) evaluated droperidol-associated risk in 45 separate studies. Twenty of these, involving 3339 patients, specifically addressed the frequency of side-effects and no cases of cardiac toxicity or sudden death were reported. Before the black-box warning in the USA, the Food and Drug Administration searched its spontaneous reporting database and the worldwide database for reports of adverse events with droperidol. It found reports of 94 deaths (14 at low dose) and 34 non-fatal cardiac events. However, droperidol was given alone in only 3 fatal cases and 3 non-fatal cases (Reference Kang, Kwon and AhnKao et al, 2003). The observation of apparently low risk with droperidol and some of the other drugs that clearly prolong the QT interval is yet to be fully explained. One possibility is their degree of penetration into the heart (Reference TaylorTitier et al, 2004).
Conclusions
Sudden death accounts for a small minority of excess deaths in people with mental health difficulties. General physical health, medical comorbidity and risk factors such as obesity, smoking, lack of exercise and diabetes have a substantial impact on accelerated mortality and no doubt influence the rate of sudden cardiac death. The role played by antipsychotic medication is not yet fully clarified. Certainly many antipsychotic drugs (including atypicals) cause a dose-dependent QTc prolongation and this is likely to be important in people who take large overdoses. Other ECG changes also occur but these have not been well studied. In large well-conducted population studies the risk conferred by QT prolongation appears to be a modest increase in mortality and sudden death. In large pharmacovigilance studies this is mirrored by an increase in sudden death in patients prescribed antipsychotics. Drugs with greater risk include thioridazine, droperidol, sertindole and ziprasidone; quetiapine, haloperidol, chlorpromazine and olanzapine can be considered of lower risk, given current evidence. The largest modifying variable for antipsychotic-induced sudden death is established (or occult) cardiovascular disease. A further consistent risk factor is the cumulative dose of antipsychotic (including polypharmacy) (Reference Trenton, Currier and ZwemerWaddington et al, 1998; Reference Harrigan, Miceli and AnzianoHennessy et al, 2002). Clinically, this is a serious concern. Reference Engdahl, Holmberg and KarlsonGanguly et al(2004) found the rate of antipsychotic combinations in 31 435 people with schizophrenia to be 40%. Comorbid physical illness should also not be underestimated: 16% of patients with schizophrenia have at least one medical condition associated with QT prolongation (Reference Jusic and LaderKalsekar et al, 2003) and up to 74% have a chronic medical condition of some type (Reference Jackson, Ditmanson and PhibbsJones et al, 2004).
In part 2 of this overview (Reference Abdelmawla and MitchellAbdelmawla & Mitchell, 2006) we will discuss monitoring of people newly prescribed antipsychotics and prevention of antipsychotic-induced sudden death.
Declaration of interest
None.
MCQs
-
1 As regards torsade de pointes and sudden cardiac death:
-
a all drugs that prolong the QTc interval produce torsade de pointes and sudden death
-
b all drugs known to cause torsade de pointes block the HERG potassium channels
-
c torsade de pointes is a form of atrial fibrillation
-
d most cases of torsade de pointes are fatal
-
e torsade de pointes must be managed by cardiac pacing.
-
-
2 As regards the QT interval:
-
a Bazett’s formula measures the QT interval
-
b QT interval represents atrial depolarisation
-
c vagal stimulation prolongs the unadjusted QT interval
-
d QT prolongation on automated readings should be checked manually
-
e shortening of QT interval is found in hypercalcaemia.
-
-
3 Epidemiological studies of antipsychotic drugs and sudden cardiac death show:
-
a dose–response relationship for certain antipsychotics
-
b that the risk of antipsychotic-induced sudden death is highest in patients taking depot antipsychotics
-
c that the risk of antipsychotic-induced sudden death is small in those without cardiac disease
-
d that sudden death typically occurs in people under the age of 30 years
-
e that sudden death does not occur in people with depressive disorder treated with atypical anti-psychotics.
-
-
4 Risk factors for torsade de pointes include:
-
a male gender
-
b hypokalaemia
-
c congenital long-QT syndrome
-
d heart failure
-
e hypomagnesaemia.
-
MCQ answers
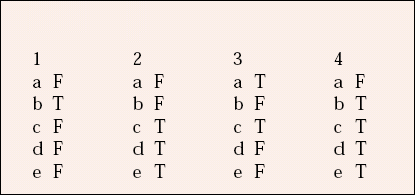
| 1 | 2 | 3 | 4 | ||||
|---|---|---|---|---|---|---|---|
| a | F | a | F | a | T | a | F |
| b | T | b | F | b | F | b | T |
| c | F | c | T | c | T | c | T |
| d | F | d | T | d | F | d | T |
| e | F | e | T | e | F | e | T |
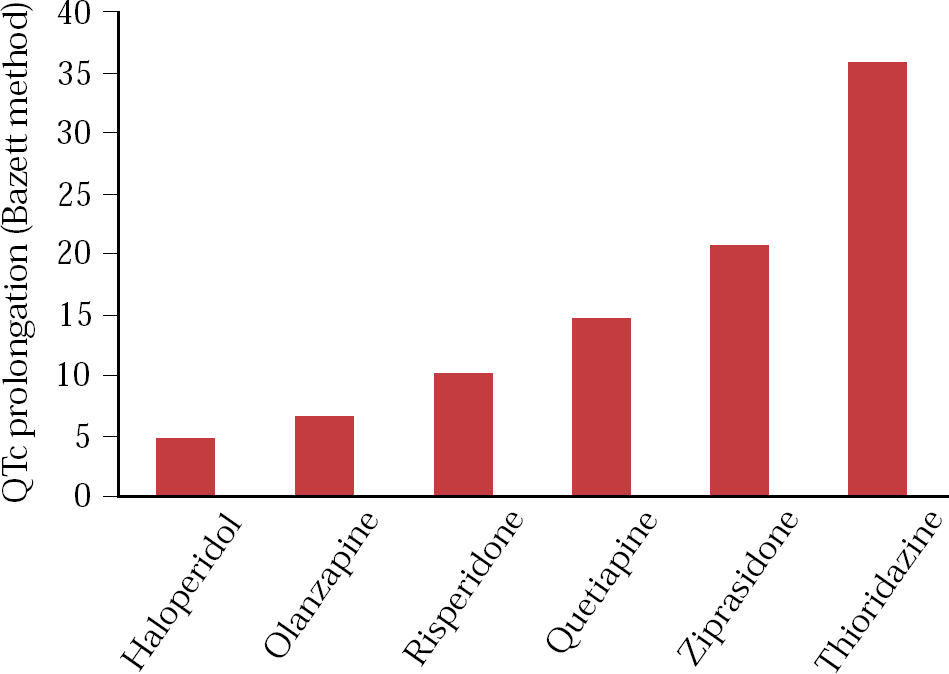
Fig 1 QTc prolongation with common antipsychotic drugs: 183 patients with normal ECGs at baseline were randomized to one of six antipsychotic drugs at maximum daily doses of: ziprasidone 160 mg, risperidone 16 mg, olanzapine 20 mg, quetiapine 750 mg, thioridazine 300 mg and haloperidol 15 mg (Data from Reference Priori, Alliot and Blomstrom-LundqvitPsychoPharmacological Drugs Advisory Committee, 2000).






eLetters
No eLetters have been published for this article.